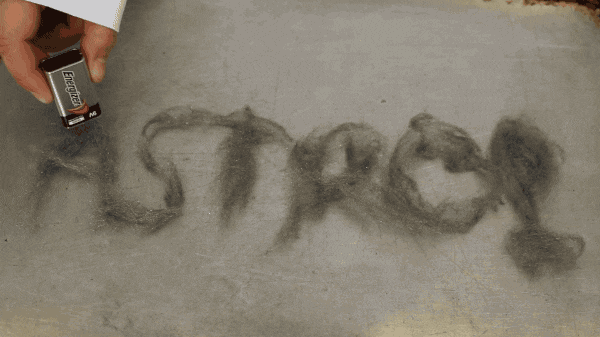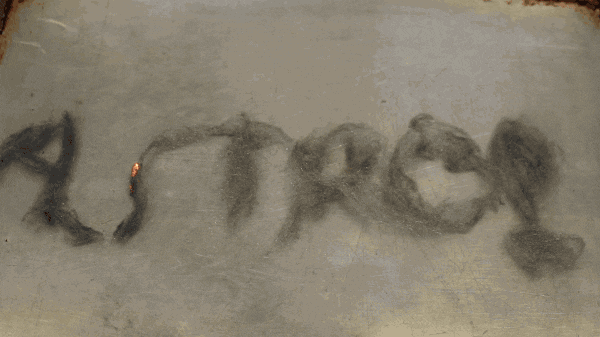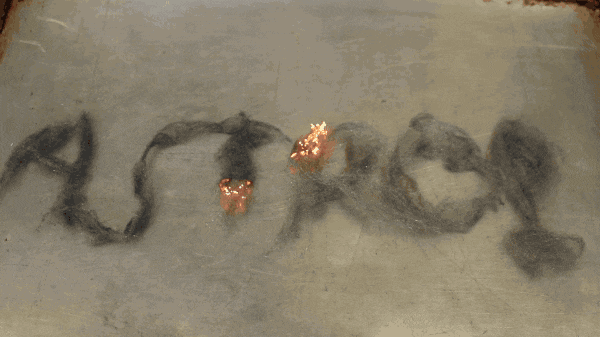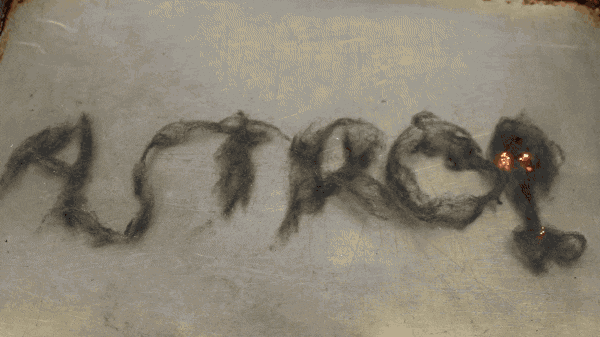You can do this experiment at home with just a few things. 9 Volt battery, steel wool, non-flammable surface, and some adult supervision.
After fluffing up the steel wool a bit place it on your non-flammable surface, then simply touch the 9 volt battery to it. It’s that simple! But what’s really happening? When you touch the 9 volt battery to it, the steel wool makes a completes the circuit of the battery.
Steel wool is made mostly of iron which is extremely conductive. The surge of electrical current from the battery heats up the iron which causes it to react with the oxygen in the air surrounding it. Electrical current is the flow of charged particles that collide with the iron atoms, slowing them down. The slowing down of the charged particles is also known as electrical resistance.
The collisions create heat. The hotter something gets, the more electrical resistance it will have. It heats up so much, that it creates a spark. The release of heat from the spark causes a new piece of iron to heat up, react, spark, release of heat… and creates a chain reaction that can continue even when you remove the battery from the steel wool.
This reaction creates iron oxide, FeO2. The hotter the steel wool gets, the more likely the oxidation reaction will happen. It can burn, unlike a nail, screw, knife, or forks because of the increased surface area ratio. There is more oxygen available in a given area for it to react with, and there can be more of a heat build up in a smaller area.