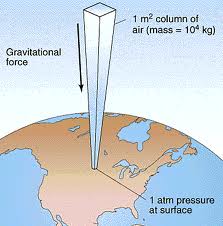
Ever wish you could control the weather? In this experiment, we’ll harness the weight of the atmosphere to make a tiny storm in a beaker! As you read this sentence, the air around you is exerting enormous pressure on your body. Imagine a jacket made of one-inch checkerboard squares. Now imagine that each tiny box weighs fifteen pounds! At sea level, atmospheric pressure is about 15 pounds per square inch. Multiply 15 by the number of square inches on the surface of your body to get an idea of how much weight you’re bearing every moment of every day.
Atmospheric pressure results from the weight of the air above you. Credit: Peter Mulroy, http://peter-mulroy.squarespace.com/air-pressure/
We don’t notice all this weight because we’re used to it. Take the pressure away, though, and awesome dynamics ensue. Heating a volume of air is one way to create a drop in pressure. Adding heat to a gas causes its molecules to speed up and spread out.
As the air inside the flask expands, some is pushed out through the tube. Invert the container into cool water and the liquid blocks the opening– the escaped air is trapped outside! The gas left inside the flask shrinks as it cools, creating a low-pressure zone in the container. Here’s where that 15 pounds per square inch comes in. Once the pressure in the flask is lowered, atmospheric pressure pushing on the surface of the water outside forces the liquid up the tube.
When the water reaches the main compartment of the container, the gas inside experiences a sudden drop in temperature. A chain reaction of cooling occurs, and water rushes in to fill the empty space left by the condensing air.