
Hydrogen: it’s the most common element in our universe, the main ingredient in stellar fusion, and the lightest element of them all. We love to play with hydrogen in the classroom because it’s highly combustible, which means it’s great for explosions! In this experiment — which is not one we recommend for home DIY — we’ll fill soap bubbles with hydrogen and light them on fire.

Hydrogen and oxygen react to form water molecules. So, how do they create such violent explosions? The water molecules have less potential energy than the sum of their hydrogen & oxygen parts, and that extra energy has to go somewhere. It’s released as light and heat! This kind of reaction is called exothermic.
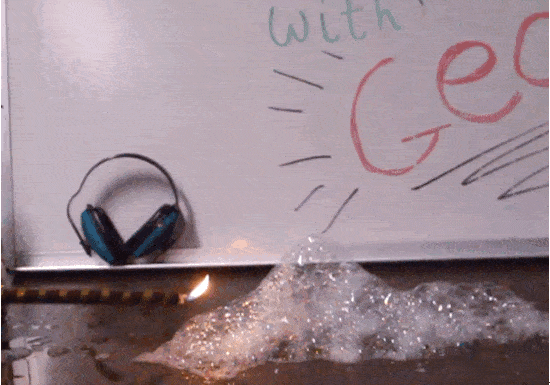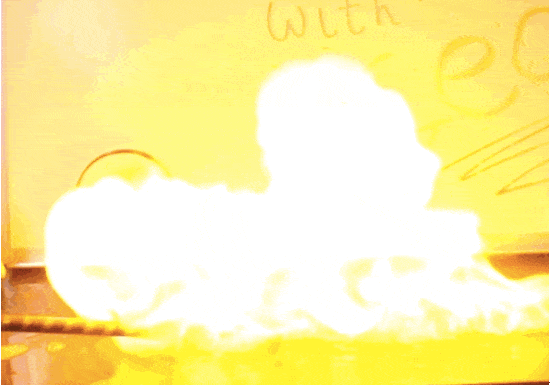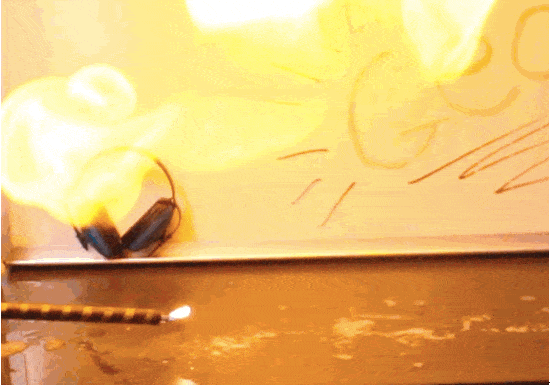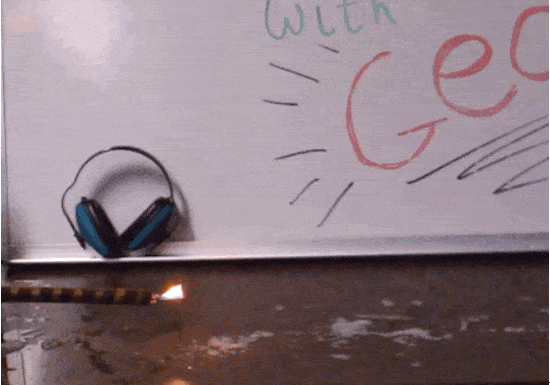
In this experiment we trap hydrogen inside of soap bubbles until we’re ready to trigger an explosion. Any container will do the trick, though, as long as it doesn’t block the activation energy. A balloon full of hydrogen creates a bigger fireball than the bubbles you see here. A single spark transforms a blimp filled with hydrogen into a massive firestorm– exactly what happened in the Hindenburg tragedy of 1937. Once the explosion is set in motion, it continues until all available fuel is consumed, for better or for worse.



